|
After the first dose, pruritic and variably painful erythematous reactions near the injection site developed in a median (range) of 7 (2-12) days after vaccine administration. Of the 16 patients, 15 developed localized cutaneous reactions after the first dose 1 patient developed a reaction only after the second dose. 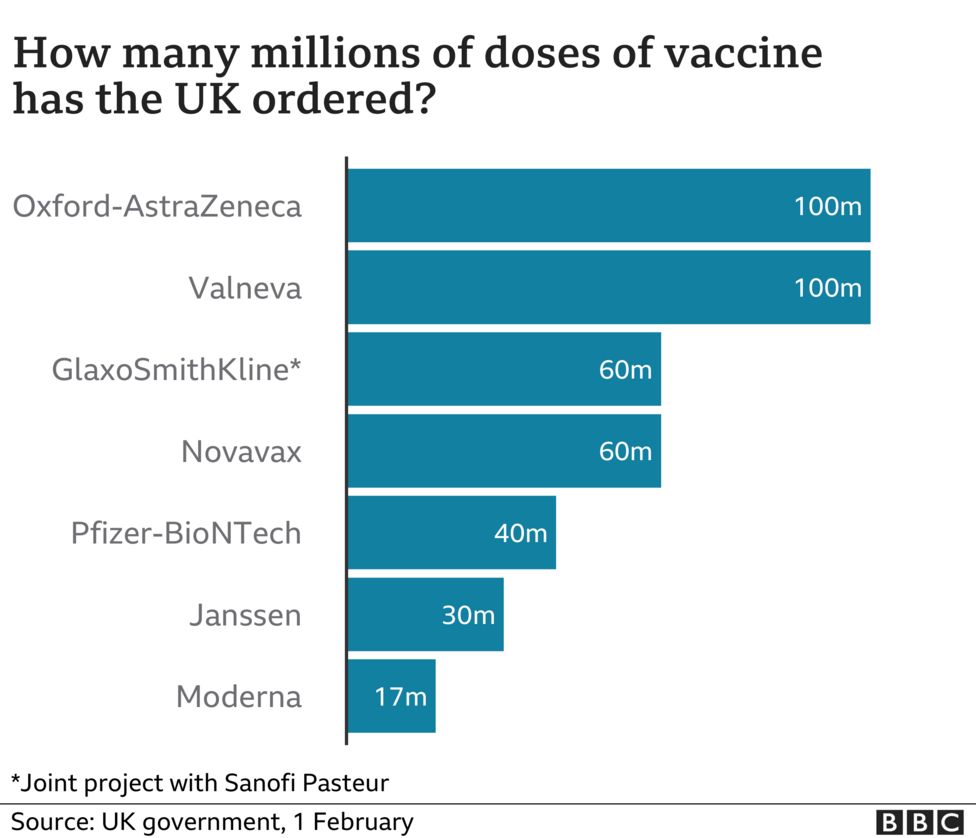
Most (13 of 16) of the patients were health care workers for the others (3 of 16), vaccine indication was for older age. All 16 participants received the Moderna COVID-19 vaccine, and only 1 reported a prior localized vaccine reaction (mild reaction to an influenza vaccine). The main characteristics of these patients with delayed cutaneous hypersensitivity reactions are shown in the Table. 
Of 16 patients, (median age, 38 years 13 women) 14 patients self-identified as White and 2 as Asian. In contrast to immediate hypersensitivity reactions (eg, anaphylaxis, urticaria), these delayed reactions (dubbed “COVID arm”) are not a contraindication to subsequent vaccination. These reactions may occur sooner after the second dose, but they are self-limited and not associated with serious vaccine adverse effects. Of participants who had a reaction to first vaccine dose (15 of 16 patients), most (11 patients) developed a similar localized injection-site reaction to the second vaccine dose most (10 patients) also developed the second reaction sooner as compared with the first-dose reaction.Ĭonclusions and Relevance Clinical and histopathologic findings of this case series study indicate that the localized injection-site reactions to the Moderna COVID-19 vaccine are a delayed hypersensitivity reaction. Results of a skin biopsy specimen demonstrated a mild predominantly perivascular mixed infiltrate with lymphocytes and eosinophils, consistent with a dermal hypersensitivity reaction. None of the participants had received the Pfizer-BioNTech vaccine. These reactions occurred at or near the injection site and were described as pruritic, painful, and edematous pink plaques. The delayed localized cutaneous reactions developed in a median (range) of 7 (2-12) days after receiving the Moderna COVID-19 vaccine. Results Of 16 patients (median age, 38 years 13 women), 14 patients self-identified as White and 2 as Asian. Main Outcomes and Measures We collected each patient’s demographic information, a brief relevant medical history, clinical course, and treatment (if any) and considered the findings of a histopathologic examination of 1 skin biopsy specimen. Objective To describe the course of localized cutaneous injection-site reactions to the Moderna COVID-19 vaccine, subsequent reactions to the second vaccine dose, and to characterize the findings of histopathologic examination of the reaction.ĭesign, Setting, and Participants This retrospective case series study was performed at Yale New Haven Hospital, a tertiary medical center in New Haven, Connecticut, with 16 patients referred with localized cutaneous injection-site reactions from January 20 through February 12, 2021. 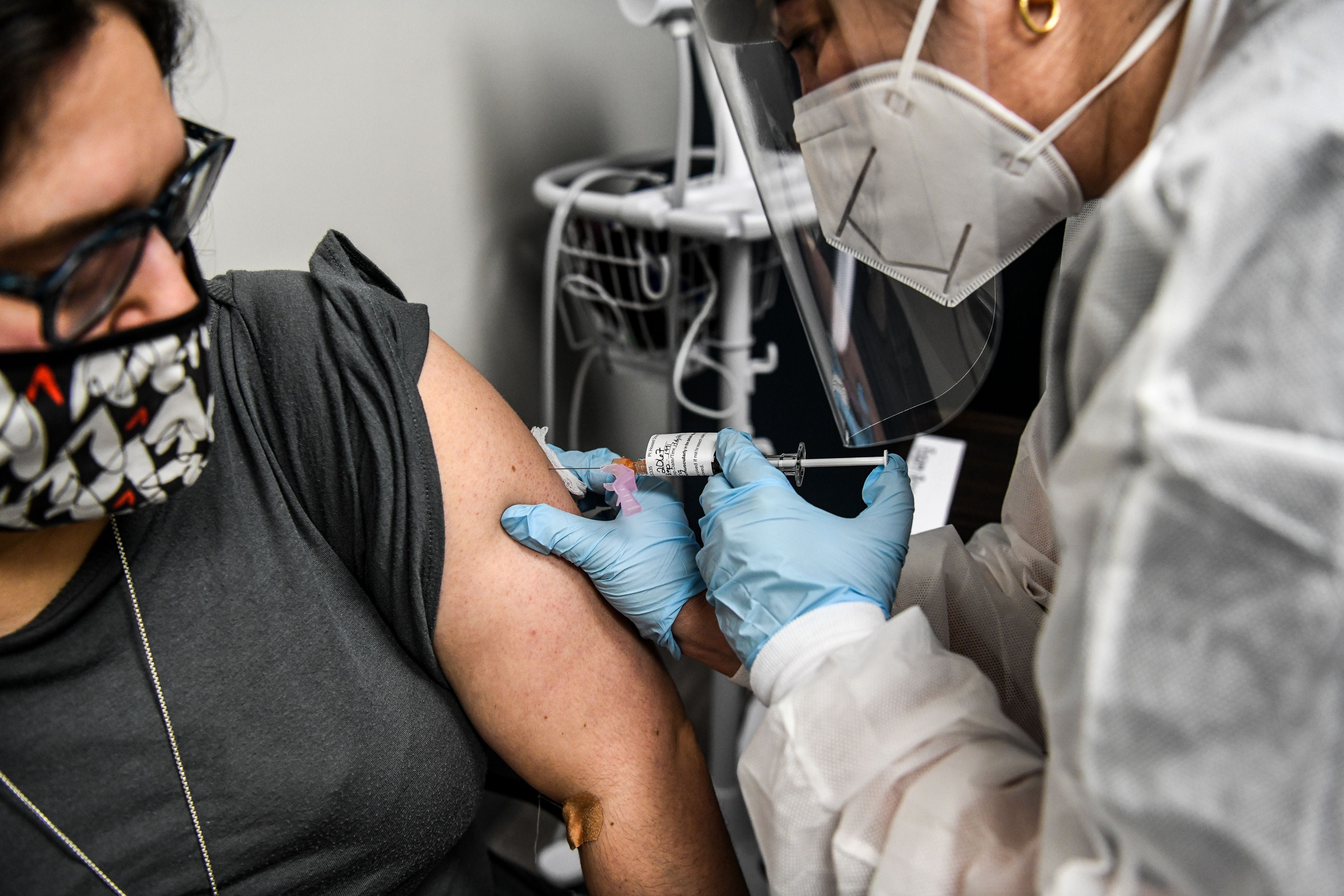
Some patients in the US have developed delayed localized cutaneous vaccine reactions that have been dubbed “COVID arm.” 
Importance In response to the coronavirus disease 2019 (COVID-19) pandemic, 2 mRNA vaccines (Pfizer-BioNTech and Moderna) received emergency use authorization from the US Food and Drug Administration in December 2020. Shared Decision Making and Communication.Scientific Discovery and the Future of Medicine.Health Care Economics, Insurance, Payment.Clinical Implications of Basic Neuroscience.Challenges in Clinical Electrocardiography.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
